 The uncertainty principle tells us that if you tried to measure its speed precisely, you would necessarily change its position-enough that you could tell whether the electron was at rest or moving with some particular speed-and thereby change its momentum. That’s such a small number, you could never measure it directly, because if you had one electron and no other particles anywhere nearby, they would be so close together that they would mutually exert forces on each other and cause the electron to change speeds. The mass of an electron is certainly much smaller than a kilogram it’s more like 9.91 × 10−31 kilograms. Thus, our new concept may provide important clues for understanding quantum-classical duality. We show that electromagnetic radiations like light (and microwaves) also exhibit a dual nature of a wave and a particle. In addition, we also extend the concept of the dual nature of matter waves (or tiny particles) from quantum physics to classical physics. However, it will not be possible to fix the position and simultaneously measure the momentum of microscopic matter waves with high accuracy.īy applying the Uncertainty Principle, we show that microscopic matter waves exhibit a dual nature of a particle and a wave. The position and momentum of macroscopic matter waves can be determined simultaneously to low accuracy. In this section, we describe the dual nature of matter waves and electromagnetic radiations. In both these formulations, Heisenberg was considering only variables. 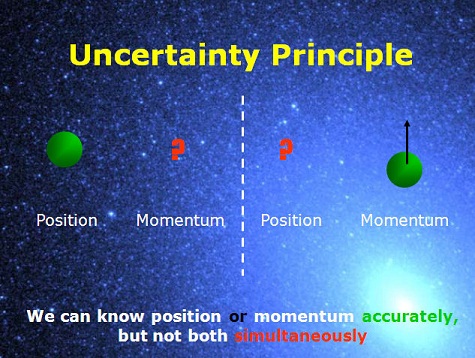
He later restated it to emphasise its mathematical nature: “The more precisely the position is determined, the less precisely the momentum is known, and vice versa”. Heisenberg originally stated his uncertainty principle as “We cannot know (or measure) both the position and the velocity of a particle at the same time”. 
Many physicists believe that it simply states an obvious fact about our inability to measure all properties accurately, rather than a fundamental limitation of nature. Some authors have questioned that interpretation and argue instead that uncertainty is a consequence of an indeterminism inherent in nature on account of quantum decoherence.Īlthough this principle was first formulated in 1927, it remains controversial. This implies that higher precision in measuring the position of a particle will result in less precision in measuring its momentum and vice versa.Ī common interpretation is that Heisenberg’s uncertainty principle limits how precisely pairs of physical properties of a particle-like its position and momentum can be known simultaneously. 
The principle states that it is impossible to measure the position and velocity with complete accuracy simultaneously. The uncertainty principle, developed by Werner Heisenberg, is one of the most widely known results in quantum physics. But now, having located it at some particular point X, we no longer know for sure where it is – our uncertainty about its location has increased! Likewise, if we try to determine its precise momentum during this time, we lose the ability to locate it. However, since momentum and energy are related, determining the former gives us information about the latter (via E=mc2) and where it is. To measure its position, we must find where the particle is at some particular time thus, we require knowledge of its momentum p. 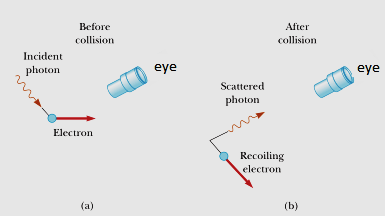
Consider a free particle that we are trying to measure: it can be represented by a wave function ψ(x, t) which acts as a probability density function. Taken at face value, Heisenberg’s uncertainty principle states that an observer can never measure both the position and momentum of an object to arbitrary precision.Īn example best demonstrates the uncertainty principle. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
